|
|
Original Article
| ||||||
| Clinical genomics: Next generation guide in difficulty-to-treat cancers | ||||||
| Iyer Sandhya1, Aarthi Ramesh1, Kavishwar Shubham1, Vijayakumar Rajeev2, Todur Seema1, Basavalingegowda Madhura1, Shafi Gowhar1 | ||||||
|
1Department of Clinical Genomics & Bioinformatics, Positive Bioscience, Mumbai, India 2Narayana Health, Mazumdar Shaw Medical Centre, Bommasandra, Bangalore, India | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar articles in PubMed] [Similar articles in Google Scholar] |
| How to cite this article |
| Sandhya I, Ramesh A, Shubham K, Rajeev V, Seema T, Madhura B, Gowhar S. Clinical genomics: Next generation guide in difficulty-to-treat cancers. Edorium J Cancer 2018;4:100006C01IS2018. |
|
ABSTRACT
| ||||||
|
Aims: Cancer as a disease has been detected to evolve through a series of genomic mutations which continue to keep accumulating depending on the aggressiveness and stage. This study aims at highlighting the significance of next generation sequencing (NGS) based genomic testing in detecting clinically actionable genomic markers in difficult to treat cancer presentations. Methods: A retrospective analysis of comprehensive NGS outcomes with attention to complete patient demographics including current and previous disease co-morbidities was performed in selected clinically significant cancer cases. Results: The study proved effective in all the 3 clinical presentations with effective treatment predictions. In Case A, presented with HER2 (ERBB2) positive breast cancer was identified with PI3K mutation proving efficient with mTOR inhibitors. Case B presented with lung adenocarcinoma also indicated the presence of JAK2 mutations. Case C who presented with pancreatic cancer was also identified with KRAS and CDKN2A mutations predicting the utility of MEK and PI3K inhibitors. Conclusion: This reiterates the need to study every cancer type as an individual case and also the significance associated with the necessity for a comprehensive analysis to detect the complex web of active biological pathway in that specific cancer case. In this research report, we present three case studies of cancer types; breast, pancreas and lung, wherein a comprehensive genomic analysis strategy was employed and its ensuing effect on designing treatment and management modalities. Our attempt is focused on highlighting the need for beyond-the-guidelines diagnosis in every cancer case for better outcomes. Keywords: Breast cancer, Genomic testing, Lung, Next generation sequencing, Pancreas | ||||||
|
INTRODUCTION
| ||||||
|
The current paradigm in clinical cancer genomics and treatment strategy is to view and treat oncogene-positive disease primarily through the lens of single hotspot oncogenic alteration. This approach does not address the potential influence of co-occurring genomic alterations present in the cancer and instead assumes single ‘driver’ as mutually exclusive from any other. Despite limited reports, the prevalence of co-occurring genomic alterations that influence clinical outcomes in advanced-stage cancers with a primary oncogenic driver is largely unknown, although recent work has suggested a potential role of several mutations. This lack of data on the prevalence and effects of multiple co-occurring genomic events exists not only for treatment-naive cancers but also for cancers that have acquired resistance to the initial targeted therapy. The diversity of genetic alterations in cancer makes it very challenging and thus the need to treat each case as novel and developing personalized diagnostic and treatment strategy has emerged as a need. Genomic testing for breast cancer has undergone a paradigm shift today from single-gene testing to assess a whole gamut of potential genomic signatures involved in the entire pathway that can affect prognosis, treatment outcome as well as metastasis and recurrence. Similarly, several genomic studies in pancreatic carcinoma have identified a set of consistently mutated genes, though the prevalence for identified targetable mutations continue to remain low. This also adversely affects the efficacy of clinical trials, due to lack of target-agents towards mutations which occur in over 20% of PDAC cases. Development of efficient therapeutic strategy for pancreatic carcinoma highlights the need for aligning clinical development with preclinical development and discovery, which has led to development of two platforms; PRECISION-Promise in the United States and PRECISION-Panc in the UK [1]. Pancreatic cancer has been proven to be the most aggressive of all cancer types with limited treatment options; in most cases surgery for survival, which again bears a poor positive outcome rate of just 20% survival for five years after pancreatic resection. Ranked as the third-leading cause of cancer-related deaths in the western world, and the 5-year survival rate being almost unchanged in the past 50 years, pancreatic ductal carcinoma (PDAC). Thus, becomes a popular subject for large scale genomic sequencing studies to identify best-suited therapeutic and management strategies [2]. Genomic studies in PDAC have identified a set of consistently mutated genes, though the prevalence for identified targetable mutations continue to remain low. This also adversely affects the efficacy of clinical trials, due to lack of target-agents towards mutations which occur in over 20% of PDAC cases [1]. Mutations in KRAS have been the most frequently identified in PDAC (in over 95% cases), but lack of successful target agents hampers treatment design [1]. Frequently mutated genes identified in PDAC include; KRAS, CDKN2A, TP53, SMAD4, TGFBR2, MLL3, SF3B1, and few studies have also identified BRAF V600E positive in 3% KRAS-negative PDAC patients [3], [4]. Development of guidelines by clinicians further reinstated the need over the hype for genetic testing, and BRCA screening along with hereditary screening panel and many such tests for different cancer types started flooding the healthcare market. A study published in the year 2010, highlighted the benefit of identifying carriers of BRCA1/BRCA2 mutation pertaining to the risk reducing salpingo-oophorectomy to lower risk of first diagnosis of breast cancer, ovarian cancer, breast and ovarian cancer mortality [5]. Identifying BRCA status is now a clinical practice even among patients as it has been shown to confer a unique phenotype and has been associated with poor outcome. Lung cancer is one of the most popular cancer types the genomic landscape of which has been well-defined to a great extent and also included in the TCGA (Cancer Genome Atlas) and a paradigm shift in treatment of NSCLC (non-small cell lung cancer) has emerged today due to possibility of use of new therapeutic options made feasible due to incorporation of genetic testing in clinical segment. The NCCN (National Comprehensive Cancer Network) guideline recommends testing for nine cancer-related genes which have therapeutic actionability. Failure to detect any targetable alteration leaves chemotherapy as the only treatment option in most cases, especially when testing is limited to few genes. The role of JAK/STAT pathway in lung cancer is also being increasingly studied as an upregulation in the same has been detected EGFR-positive as well as EGFR-resistant NSCLC apart from its role as a mediator in lung cancer metastasis [6]. In such cases, use of JAK2 inhibitors has been detected to re-sensitize cells towards EGFR TKIs [7]. Studies have also identified a particular JAK3 mutant protein with p.V722I to activate and promote PD-L1 expression [8]. Clinical genomics holds the key to meeting many of the global healthcare challenges of the years ahead. To close the gap between translational research, clinical diagnosis and treatment, the healthcare and research community is working towards the integration of genomic data into clinical workflows. In the last few years, cost for genomic testing have plummeted, as advances in sequencing technology have made individual genome sequencing economically feasible. In this study, we present three cases of solid tumors at different disease stages, subjected to molecular analysis to identify the underlying genomic prognostic factors as well as to recommend best-suited therapeutics for disease management. | ||||||
|
MATERIALS AND METHODS
| ||||||
|
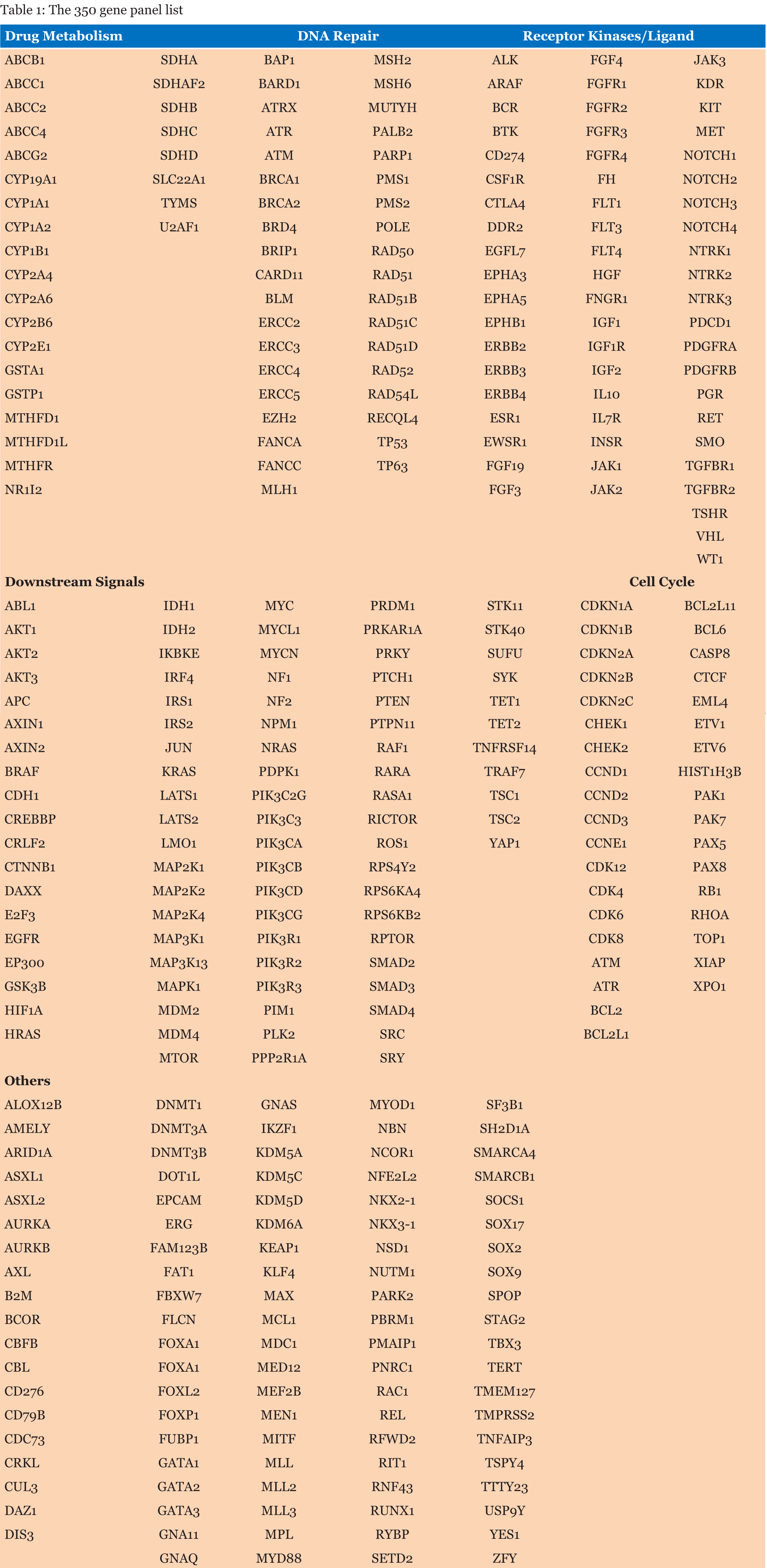
All the experiments were carried out according to the appropriate guidelines and are approved by the PositiveATGC’s (ACADEMY for TRAINING in GENOMICS and CLINICAL APPLICATION) scientific review committee. A written informed consent was acquired from all subjects as per the protocol of review committee for the utilization of their clinical samples in this study. Targeted sequencing of 350-cancer related genes was done on ctDNA/FFPE from the patients using the Illumina Next-seq platform. The target region included >5000 coding exons of canonical transcript isoforms, >100 exons of noncanonical transcripts, as well as 35 introns of 15 recurrently rearranged genes. In addition, the panel included probes to capture a 100-bp region in the TERT promoter. The bioinformatics analysis of the raw data was done using our trademark TEST pipeline to call out all four categories of genomic variants; SNVs, Indels, CNVs and Structural variants. Clinically relevant genomic alterations were defined as genomic alterations associated with response to therapies currently available or in target-driven clinical trials. The list of all the 350 genes tested has been shared in Table 1. | ||||||
RESULTS | ||||||
|
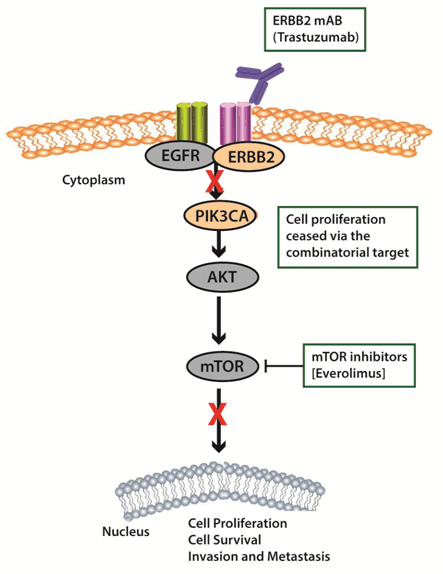
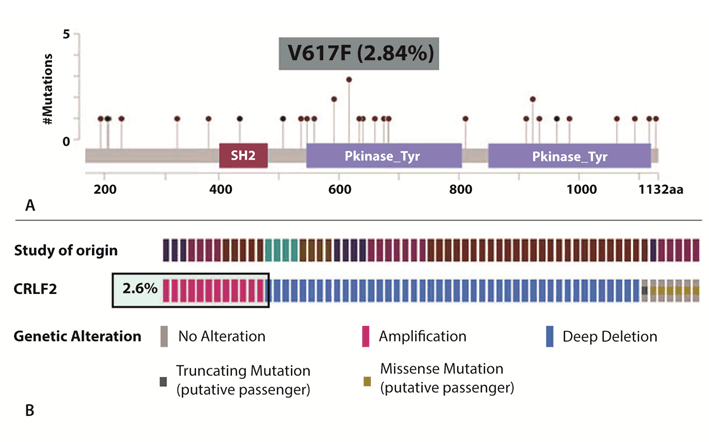
Clinical presentation Targeted NGS assay test designed for use in clinical oncology is routinely prescribed to identify personalized treatment plans as well as design future management strategies for all types of solid tumors in different stages. For this case study, we present analysis and further outcome for three different cancer cases. A 38-year-old female diagnosed with breast cancer in 2014, exhibiting symptoms of recurrence with metastasis (Case A). A 71-year-old male diagnosed with progressive lung carcinoma and bone marrow myelofibrosis in the year 2016, and exhibiting progressive disease with metastasis (Case B). A 42-year old male diagnosed with stage IV pancreatic cancer in the year 2016, and exhibiting progressive disease with lung metastasis (Case C). Genomic analysis and Treatment outcomeCase A – Use of mTOR inhibitors with anti HER2 agents: The patient was 38-year-old female affected by stage 3A HER2 positive breast cancer; specifically ductal carcinoma of the left breast. Before being recommended for the genomics test, the patient had undergone a surgical intervention involving radical mastectomy as well as chemotherapy involving platinum agents, vinorelbine, cyclophosphamide and trastuzumab. The patient had suffered recurrence and the cancer had metastasized in the lung and T11 sternum vertebrae. Comprehensive genomic analysis identified two key pathogenic variants in PIK3CA, c.3140A>G which leads to an amino acid change p.His1047Arg (H1047R) and a variant in TP53 gene, c.772G>A which causes an amino acid change p.Glu258Lys (E258K), apart from confirming her2 (ERBB2) amplification. Deregulation of the PI3K (phosphatidylinositol 3-kinase) pathway either due to mutation in the alpha subunit or loss of PTEN function has been associated with various cancers, and studies have identified PIK3CA mutations to be associated with overexpression of ERBB2, expression of hormone receptors; ER (estrogen receptor) and PR (progesterone receptor) as well as lymph node metastasis [9]. The TP53 variant has been reported to have been identified in an individual with early-onset breast cancer and family history criteria for Li-Fraumeni syndrome and also another individual affected by early onset-gastric and brain cancer with a family history of gastric cancer [10], [11]. Identification of ERBB2 amplification also opens up avenues to use target agents, and based on identification of co-presence of PIK3CA and ERBB2 variation, a combination of mTOR inhibitors with anti HER2 was postulated to work better for the patient to successfully tackle the entire prognostic machinery which is also presented in Figure 1. The patient follow-up indicated good response to the combinations suggested. Case B – Use of JAK inhibitors: A 71-year-old male affected by adenocarcinoma of the right upper lung along with a condition of bone marrow myelofibrosis. Before being recommended for the targeted NGS testing, the patient had undergone surgical intervention involving removal of gall bladder and a prostate surgery. When recommended for genetic testing, the disease stage was reported to be progressive with lung metastasis and a prior genetic testing reported the patient to be negative for EGFR, ALK and ROS1. The current testing objective was to identify factors associated with disease progression and also directives for future treatment plans. Our 350-cancer gene targeted NGS panel testing done on ctDNA extracted from the blood sample submitted by the patient, identified two pathogenic alterations involving a mutation in JAK2 and an amplification in CRLF2. The JAK2ClinVar pathogenic/likely pathogenic variant identified involves a change c.1849G>T, leading to an amino acid change of p.Val617Phe (V617F) and has been associated with polycythemia vera and myeloproliferative disorders among others. In case of amplification in CRLF2, studies have identified such a presence to increase activation of JAK/STAT pathway culminating in increased proliferation of cancer [12]. Aberrant signaling of JAK/STAT pathway has also been reported in hematological malignancies, and such an identification in this particular case becomes relevant as the patient had been reported to be affected by bone marrow myelofibrosis [13]. A meta-analysis indicating the statistics of JAK2 and CRLF2 alterations in lung cancer is presented in Figure 2 [A and B]. The occurrence of JAK2 p.V617F mutation constituting 2.84% of the overall occurrence of JAK2 alterations in lung cancer cell line studies. The statistics of CRLF2 alterations in 2.6% of lung cancer cases as per combined lung cancer studies [http://www.cbioportal.org/] . For disease management, use of JAK1/JAK2 inhibitors wererecommended to tackle the prognosis by the JAK/STAT pathway as well as CRLF2 amplification. The follow-up with the treating physician indicated the patient to be responding well to use of the JAK inhibitor recommended in our report. Case C – Use of MEK and PI3K inhibitors along with immunotherapy: A 42-year-old male diagnosed with pancreatic adenocarcinoma in stage 4 (T4N1M1). The disease condition was identified to be progressive and the cancer had metastasized in the lungs. No surgical or radiation intervention had been done and the patient was treated with chemotherapeutics when recommended for the targeted-NGS genomic testing. The patient had been treated with FOLFIRINOX, gemcitabine as well as paclitaxel which had been discontinued and started on bevacizumab, erlotinib and luporal. The objective of recommending the NGS-test involved identification of genetic mutations which could influence disease prognosis as well as treatment outcome. The 350-cancer gene targeted NGS panel matched-normal testing done on ctDNA extracted from the blood sample submitted by the patient, identified two somatic actionable alterations in KRAS and CDKN2A genes. Additionally, the patient was detected to be positive for microsatellite instability. The identified ClinVar pathogenic/likely pathogenic variation in KRAS includes the c.35G>A which causes a change designated as p.Gly12Asp (G12D) and has been designated as a key actionable oncogenic alteration in colorectal cancer and NSCLC. The other ClinVar pathogenic variant identified in CDKN2A gene; c.131dupA is an insertion variant which leads to frameshift termination, p.Tyr44Terfs and has been associated with hereditary cancer predisposing syndrome. Though KRAS and CDKN2A genes have been included as relevant to pancreatic adenocarcinoma in the NCCN guidelines, their inclusion while selecting for single-gene analysis or key cancer genetic alteration analysis is very rare. Identification for presence of microsatellite instability also further opens up avenue for immunotherapy use in a clinical setting. We recommended the use of combination of MEK and PI3K inhibitors along with immunotherapy agent to show benefit. The follow-up indicated the patient to be doing well as of writing this manuscript. | ||||||
| ||||||
| ||||||
DISCUSSION | ||||||
|
Knowledge of co-existent actionable genomic alterations may increase prospective treatment choices. Our case reports highlight the need for comprehensive genomic based analysis over the traditional single-gene testing strategies to expand the arena of treatment choices based more on active pathways than specific cancer type. In case of the breast cancer, the identified pathogenic mutation; p.H1047R in the kinase domain of PIK3CA occurs in the highly conserved region and thereby increases the catalytic activity of the protein in-turn leading to enhanced downstream activation and oncogenic progression [14]. With reference to considering the detected TP53 p.E258K mutation, functional studies have identified this mutation to affect transcriptional activation, and is tagged as a non-conservative amino acid substitution change. This change is also important as it occurs in the conserved DNA binding domain [15], [16]. Further the patient was detected to be positive for Her2/ERBB2 amplification, and this finding could be corroborated with the fact that the patient had been treated with 12-cycles of trastuzumab and had exhibited response. However, the identification of the PIK3CA mutation through our comprehensive analysis highlights the presence of a downstream variation apart from a receptor aberration, indicating the possible inefficacy of trastuzumab alone, ending up in development of recurrence. Thus, for effective disease management, recommendation for combination agents were done; like everolimus, an mTOR inhibitor with trastuzumab. Studies have also identified benefit of a combination of everolimus with fulvestrant for recurrent breast cancer, and hence the same was also recommended [17]. The detection of the TP53 loss-of-function mutation further highlights poor outcome with platinum agents and we recommended avoidance to the same to reduce incidences of toxicity due to cellular apoptotic inefficacy. Analysis of the lung adenocarcinoma case with bone marrow myelofibrosis, identified the JAK2 pathogenic variant, p.V617F documented to disrupt the auto-inhibition of JAK2 resulting in constitutive activation of downstream signaling through the JAK/STAT, MAPK and the PI3K/AKT pathways, providing proliferative advantage to tumor cells [18], [19] . Studies have identified frequency of JAK2 mutations to be roughly 1% in lung cancer. A comprehensive meta-analysis of 1020 samples in varied cancer types yielded 2.84% of JAK2p.V617F mutation in samples of hematopoietic and lymphoid tissue. Individual sample studies also indicate JAK/STAT pathway alterations to be reported in hematological malignancies. Amplifications in CRLF2has been well established in leukemia, and a recent meta-analysis including 3484 lung cancer cases detected the frequency of the same to be 2.6%, indicating significance [20], [21], [22], [23], [24]. Identification of both the JAK2 and CRLF2 variation through a comprehensive analysis increases the possibility for use of targeted therapeutics even in the absence of lung cancer specific alterations due to presence of the myelofibrosis condition. These findings strongly suggest JAK inhibitors like ruxolitinib, an ATP-binding competitive inhibitor of both JAK1 and JAK2 could be beneficial in inhibiting the proliferation of JAK2 p.V617F cells in myelofibrosis, post-polycythemia vera myelofibrosis, and post-essential thrombocythemia myelofibrosis as has been shown earlier [18] . Further on, an ongoing phase II clinical trial initiated to study the efficacy of ruxolitinib with chemotherapy in NSCLC (NCT02119650) could be another option. The co-occurring CRLF2 amplification aggravates the functional imbalance of JAK/STAT signaling, and thus was postulated to be targeted by ruxolitinib which has been proven clinically beneficial in many studies [12]. Though lung cancer relevant alterations such as EGFR, ALK, etc., were identified negative in our analysis, few additional findings assisted in making available better targeted therapeutics since the patient holds myelofibrosis conditions. On further follow-up post recommendations, the disease was reported to be stable in the patient after treatment with ruxolitinib. In case of the third case study involving pancreatic adenocarcinoma, the KRAS pathogenic variant identified, p.G12D has been documented to increase proliferation, invasiveness as well as differentiation properties by increased constitutive activation of the RAS GTPase. Further, the identified CDKN2A insertion mutation involving codons 131 and 132 leads to generation of a premature stop codon leading to protein truncation or non-sense mediated mRNA decay. This event has been postulated to cause improper inhibition of CDK4/6 resulting in aggravated cell proliferation. When it comes to recommendation of therapeutics, few limited case studies have highlighted the use of MEK inhibitors among KRAS p.G12D positive pancreatic adenocarcinomas [25]. Most of the reported cases had significant – at times exceptional responses justifying overall recommendations (including NCCN guideline) to offer MEK and PI3K inhibitors, such as single-agent trametinib, selumetinib, buparlisib or a combination of pembrolizumab with trametinib and dabrafenib. Trametinib/selumetinib is a reversible, allosteric inhibitor of MEK1 and MEK2 activation and of MEK1 and MEK2 kinase activity. These have also been shown to bear a positive effect on cancers with BRAF and KRAS active downstream signaling pathway as these result in the constitutive activation of the MAPK pathway [26], [27], [28]. The identified KRAS mutation since can activate downstream signaling pathways thus anti-EGFR (cetuximab) and TKIs (gefitinib and erlotinib) may not prove beneficial. PI3K inhibitors are also being effectively studied in various solid tumors [29]. Knowledge of coexisting actionable genomic alterations may increase prospective treatment choices. It has been shown MEK inhibitors effectively cooperate with CDK4/6 inhibition in eliciting cell cycle exit, based upon which two combination therapies also had been further recommended; palbociclib with nab-paclitaxel as per a phase I clinical trial in metastatic pancreatic ductal adenocarcinoma (NCT02501902) and second one being palbociclib with cisplatin/carboplatin as per a phase I clinical trial in advanced solid malignancy cases (NCT02897375). Thus using clinical genomics, the medical community has been able to carve out precision-based treatments by making sense of vast amounts of available clinical genomics data, ultimately improving the lives of millions of people. Several studies also present the utility of nanomaterials in cancer therapy through genomic testing [30], [31]. Our case studies also emphasize the significance of a comprehensive genomic analysis in identifying active pathways and then further targeting the same to attain better disease control. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Iyer Sandhya – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Aarthi Ramesh – Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Kavishwar Shubham – Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Vijayakumar Rajeev – Substantial contributions to conception and design, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Todur Seema – Analysis and interpretation of data, Drafting the article, Final approval of the version to be published Basavalingegowda Madhura – Acquisition of data, Analysis and interpretation of data, Drafting the article, Final approval of the version to be published Shafi Gowhar – Substantial contributions to conception and design, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of Submission
The corresponding author is the guarantor of submission. |
|
Source of Support
None |
|
Consent Statement
Written informed consent was obtained from the patient for publication of this study. |
|
Conflict of Interest
Author declares no conflict of interest. |
|
Copyright
© 2018 Iyer Sandhya et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|