| Table of Contents |  |
|
Editorial
| ||||||
| Drug repositioning for cancer therapy: Overview of my current research | ||||||
| Sungpil Yoon | ||||||
|
1Breast & Endocrine Cancer Branch,Research Institute, National Cancer Center, South Korea.
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Yoon S. Drug repositioning for cancer therapy: Overview of my current research. Edorium J Cancer 2015;1:8–11. |
|
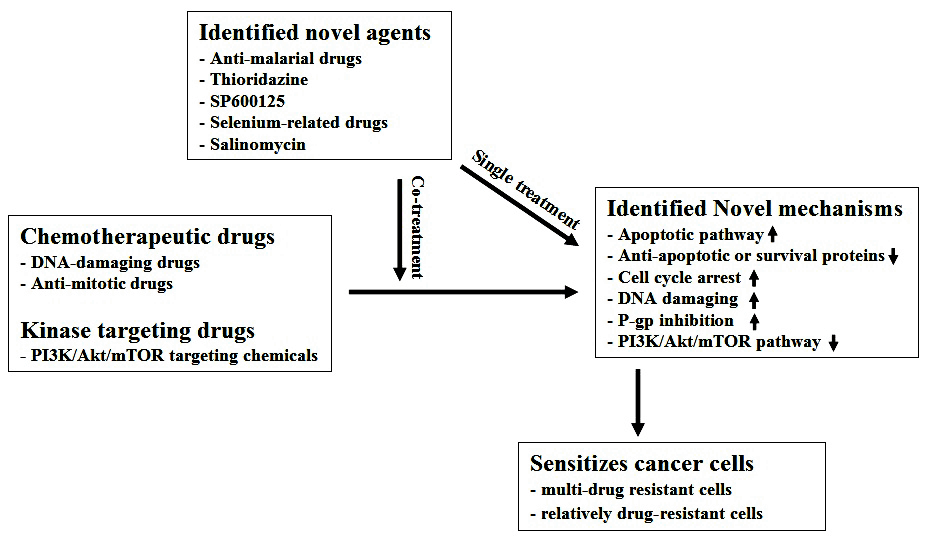
Chemotherapy is widely used for the treatment of various cancers. DNA-damaging and anti-mitotic drugs are the two types of chemotherapeutic drugs. DNA-damaging drugs such as doxorubicin, etoposide, and cisplatin, are involved directly in DNA double strand breaks. Anti-mitotic drugs inhibit mitosis by targeting microtubules and preventing their polymerization or depolymerization. Paclitaxel, docetaxel, vincristine, vinorelbine, vinblastine, and halaven are some examples of anti-mitotic drugs. While chemotherapeutic drugs are widely used to treat cancer, patients develop resistance to these drugs, specifically after prior exposure to chemotherapy. One of the important resistance mechanisms involves increased expression of P-glycoprotein (P-gp) on the cancer cell membranes to pump-out the chemotherapeutic drugs, in an attempt to avoid drug-induced toxicity. Identifying sensitizing mechanisms or sensitizing drugs for these resistant cancer cells would contribute to a better understanding of the mechanism of drug-induced resistance treatments. For example, it would be beneficial if a drug could inhibit P-gp expression as well induce cellular apoptosis. Such drugs can be effective not only for single treatment and but also show increased efficacy in combination with other chemotherapeutic drugs. In addition, identification of both non-P-gp substrates and toxic drugs would be a prominent contribution for treating resistant cancer. Drug repositioning or drug repurposing is the application of known drugs to new indications. It has been used for various diseases and is inexpensive, avoidance of time-consuming process of a significant number of toxicity tests are a few advantages of drug repositioning. It is a powerful tool to identify effective treatment options for new diseases in a timely manner. For example, the urgent need for pharmacological treatments for resistant cancer can be efficiently addressed with drug repositioning and can be applied to human patients at a relatively faster pace. Successful application of antiangiogenic drug, thalidomide for cancer treatment is an example of drug repositioning. Experimental methods such as high-throughput screening of various drug collections, including repositioned drugs already in clinical use, and literature-guided search can be used to identify repositioning drugs. The candidate repositioning drugs can be also applied in combination with chemotherapeutic drugs, provided, the efficacy of the combination is established. Based on experimental candidate approaches guided by literature search, our laboratory has tried to identify novel targets to treat resistant cancer with repositioned drugs already in use. We hypothesized that the repositioned drugs can be used for patients resistant to conventional treatment. This study aimed to identify the novel mechanisms of repositioning drugs, sensitize resistant cells, inhibit P-gp expression, or improve the efficacy of repositioned drugs when used in combination with chemotherapeutic drugs. Another prominent goal of our lab was to identify best derivative of the repositioned drugs. Taken together, the purpose of this study was to identify the mechanisms underlying the sensitizing effects of repositioned drugs against resistant cancer cells. Five findings of our laboratory have been described below and summarized in a brief diagram (Figure 1). 1. Co-treatment with the anti-malarial drugs mefloquine and primaquine highly sensitizes drug-resistant cancer cells by potentiating P-gp inhibition. Atovaquone, chloroquine, primaquine, mefloquine, artesunate, and doxycycline are the most commonly used anti-malarial drugs. These anti-malarial drugs are also shown to be potentially useful in the treatment of cancer. Results and Discussion: With an exception of doxycycline, all the other anti-malarial drugs tested could sensitize both sensitive KB and resistant KBV20C cancer cells to a similar extent, suggesting that anti-malarial drugs could be used for sensitive as well as resistant cancer cells. Primaquine and mefloquine, highly sensitized KBV20C-resistant cells to vinblastine treatment. However, primaquine- or mefloquine-induced sensitization to vinblastine treatment was not observed in sensitive KB parent cells, suggesting that the observed effect is specific to resistant cancer cells. We demonstrated that the primaquine and mefloquine sensitization mechanism mainly depends on the P-gp inhibition. 2. Thioridazine specifically sensitizes drug-resistant cancer cells by inducing apoptosis and P-gp inhibition. Thioridazine was originally used to treat patients with psychosis. Thioridazine's cancer-sensitizing ability was demonstrated in various cancer models. Thus, thioridazine is currently considered as a potential anti-cancer drug and can be administered during chemotherapy. | ||||||
| ||||||
|
RESULTS AND DISCUSSION
| ||||||
|
Thioridazine was found to markedly sensitize drug-resistant KBV20C cancer cells compared to their effects on sensitive KB parent cells. Thioridazine prominently increased apoptosis specifically in KBV20C cells, suggesting that the difference in sensitization between the resistant and sensitive cells can be attributed to the ability of thioridazine to induce apoptosis. Thioridazine could also inhibit P-gp activity in the resistant KBV20C cells. These observations suggest that the mechanisms underlying thioridazine-induced sensitization in resistant KBV20C cells involve both apoptosis and P-gp inhibition. In addition, we also observed that co-treatment with thioridazine and vinblastine induces increased sensitization in KBV20C cells than that in KB cells. 3. SP600125 overcomes antimitotic drug-resistance in cancer cells by increasing apoptosis independent of P-gp inhibition. The JNK inhibitor SP600125, a mimetic for Müllerian-inhibiting substance, was found to be effective against doxorubicin-resistant cells in an in vivo mouse model. Co-treatment with doxorubicin and SP600125 sensitized cancer cells. Other studies have described various mechanisms underlying SP600125's cancer-sensitizing ability. Thus, SP600125 has potential as an anti-cancer drug. While SP600125 is not a therapeutic candidate for humans, derivatives of SP600125 can be used, once the specific features or mechanisms underlying its sensitizing effects are identified. Results and Discussion: SP600125 sensitized the antimitotic drug-resistant KBV20C cancer cell line. Interestingly, SP600125-induced markedly sensitized resistant KBV20C cancer cells compared to their effects on sensitive KB parent cells. The mechanism of SP600125-induced sensitization involved G2 cell cycle arrest. KBV20C cells treated with SP600125 and anti-mitotic drugs were sensitized more than the cells treated with SP600125 alone. This suggests that SP600125 can restore the sensitization of resistant cancer cells to anti-mitotic drugs. The sensitization mechanism was observed to be independent of P-gp inhibition. 4. Selenate specifically sensitizes drug-resistant cancer cells by increasing apoptosis via G2 phase cell cycle arrest independent of P-gp inhibition. Clinical trials for selenium-related drugs have been designed to validate their efficacy as chemopreventive agents. In addition, selenium-related drugs have been shown to inhibit tumorigenesis in various experimental models including cell lines, mouse, and rats. Results and Discussion: Among the selenium-related drugs, selenate, selenite, selenomethionine, methylselenocysteine, and methaneselenic acid are the most commonly studied selenium-derived drugs to treat cancer in humans. The sensitizing effects of all the five drugs on KBV20C cells were comparable to their effects on sensitive parent KB cells, suggesting that selenium-derived drugs can be used for drug-resistant cancer cells. We also observed that these drugs did not inhibit the P-gp pumping-out ability in resistant KBV20C cells. Interestingly, we observed that selenate highly sensitized drug-resistant KBV20C cells by activating the apoptotic pathway. In addition, selenate-induced toxicity was associated with increased G2-phase cell cycle arrest in KBV20C cells, suggesting that the selenate-induced increase in apoptosis was a consequence of cell cycle arrest. 5. Co-treatment of salinomycin sensitizes chemotherapeutic- or Akt-targeting drug-treated cancer cells by increasing apoptotic cell death. Salinomycin was originally used to eliminate bacteria, fungi, and parasites. More recently, this drug has been exploited to inhibit the growth of tumor stem cells and is considered a potential anticancer drug. Mechanism of salinomycin sensitization is under investigation. In addition, various synergistic studies of salinomycin sensitization mechanism are also being investigated. | ||||||
|
RESULTS AND DISCUSSION
| ||||||
|
Salinomycin sensitizes doxorubicin-, etoposide-, radiation-, or anti-mitotic drug-treated cancer cells to apoptosis by inducing DNA damage and downregulating p21 protein levels via increased proteasomal activity. Two Akt-targeting drugs (MK-2206 and AZD5363) in combination with salinomycin were evaluated for their sensitizing effects on cancer. Co-treatment with MK-2206 and salinomycin sensitizes cancer cells by reducing both pAkt and total Akt levels. Co-treatment with salinomycin also sensitizes AZD5363-treated cancer cells to apoptosis. | ||||||
|
Conclusion
| ||||||
|
These drugs are already used clinically or have passed preclinical trials (exception of SP600125) and therefore, are ideal therapeutic candidates, provided, their mechanism of action on resistant cancer is completely understood. These drugs could be used without further toxicity evaluation. My study would be a major contribution to improve the efficacy of various combination chemotherapeutic treatments for cancer patients who develop resistance to chemotherapeutic drugs. | ||||||
|
Abbreviations:
| ||||||
|
P-glycoprotein (P-gp) Keywords: Current research, Drug repositioning, Resistant cancer | ||||||
|
Acknowledgements
| ||||||
|
This work was supported by a research grant (NCC1310210) from the National Cancer Center, South Korea. | ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Sungpil Yoon – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2015 Sungpil Yoon. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|